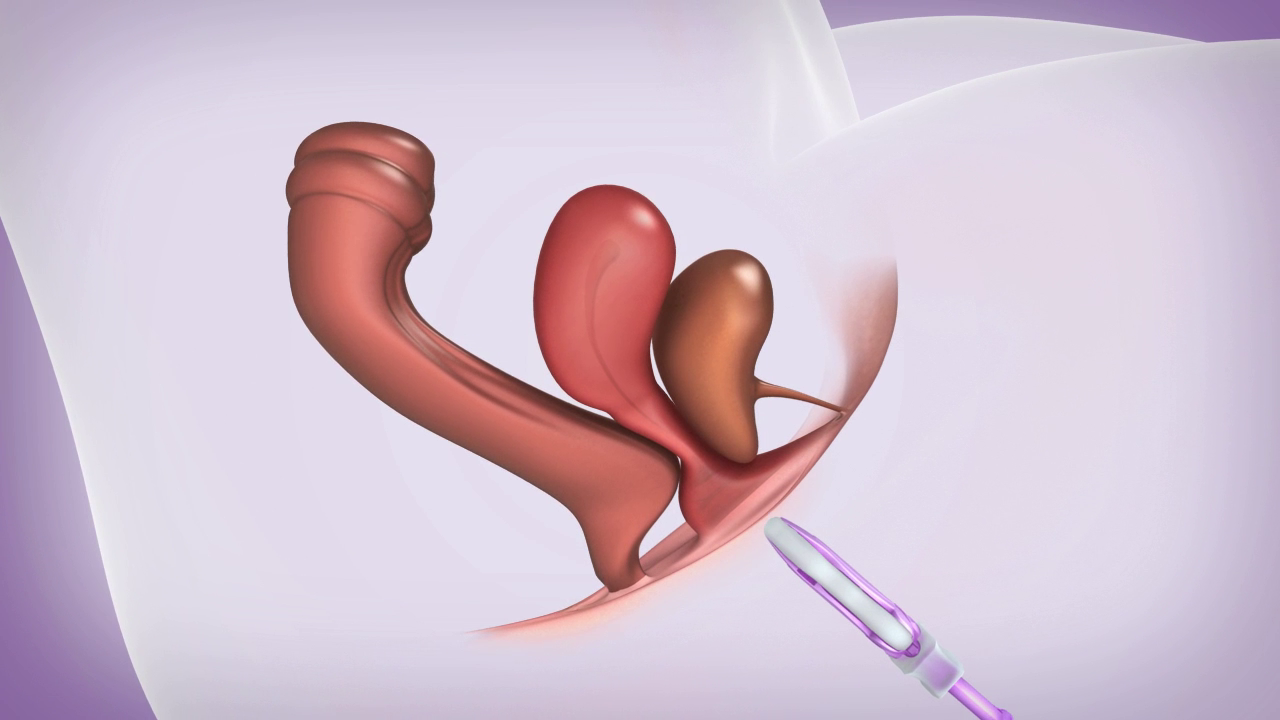
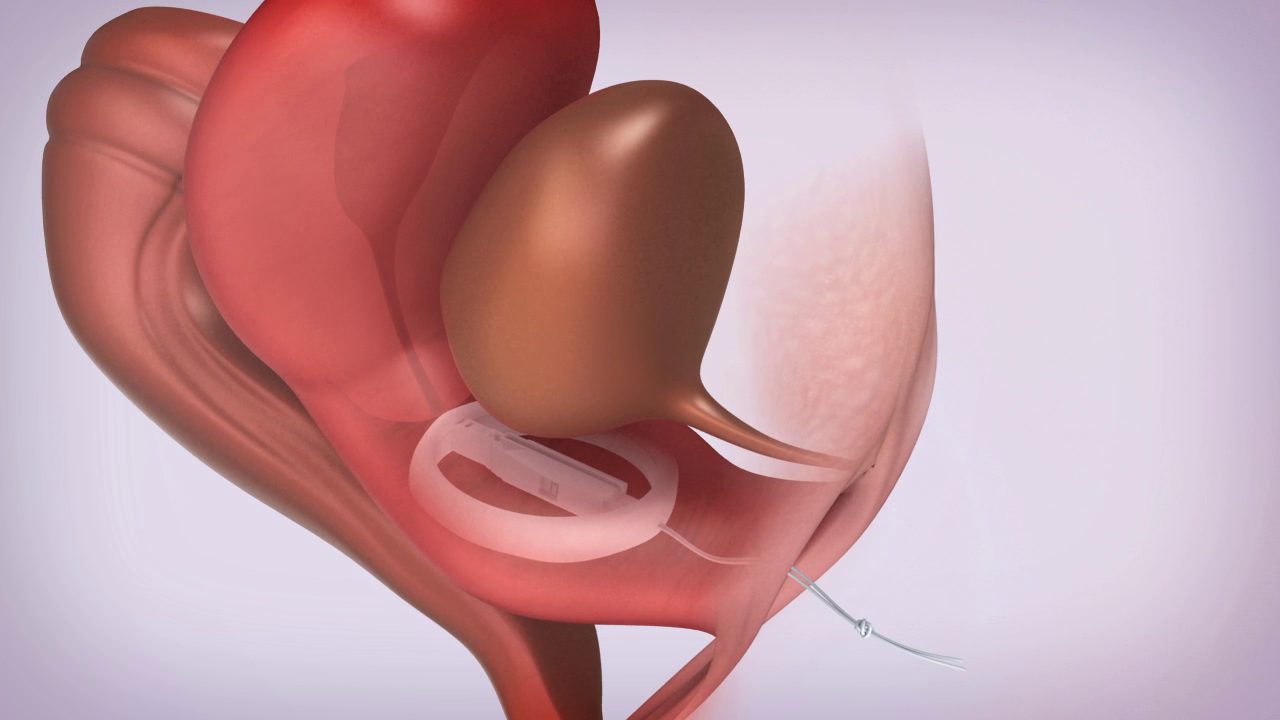
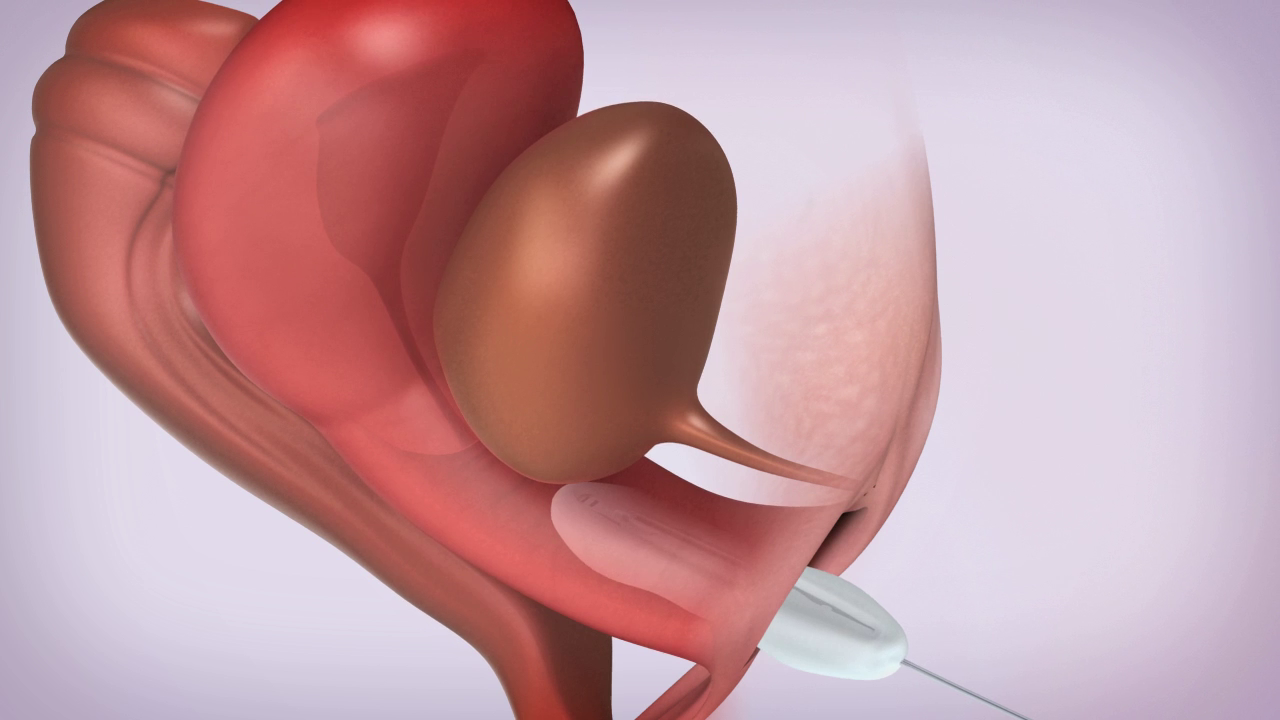
A User Friendly Device
All ConTIPI Medical’s products for various Pelvic Floor Disorders in women are designed to be user friendly and different from existing devices available in the market.
All products are considered to be non-invasive vaginal devices. They are all disposable, used for a predefined, short period of time, with no need for cleaning between uses, thus avoiding the need to insert a reusable device.